- Septoria tritici
- Zymoseptoria tritici leaf spot Jon West
- Septoria Jon West2019b
- Septoria Jon West2019
- Septoria4 Jon West
- Septoria3a Jon West
Damage
Septoria leaf blotch is the most important foliar disease of wheat in the UK. It is caused by Zymoseptoria tritici (previously known as Mycosphaerella graminicola and by the previous name for the asexual-stage, Septoria tritici). The disease is initiated by wind dispersed ascospores, which are released continually from crop debris, in the autumn to early winter and again in late spring-early summer (Hunter et al. 1999). In addition, the disease is intensified and dispersed onto newly emerging leaf layers by rain-splashed conidia (asexually-produced spores). This means that the crop is continually exposed to infection, except during dry periods when both types of the spores are unable to infect successfully. As a result, studies performed years ago to assess the economics and effectiveness of different fungicide applications, performed over different growth stages in experimental plots, found that the economic strategy was to protect the final three leaves (leaves 3, 2 and flag leaf). Autumn or winter applications of fungicide were not effective due to the continual re-exposure of the crop to infection by either ascospores or conidia. In the early 2000s, when wheat prices were very low (£75/tonne), experiments at Rothamsted found the most cost-effective regime was simply a single flag-leaf fungicide application. As the value of the crop increased, it became cost-effective to spray at least twice in spring, even more so if spring weather is unusually wet.
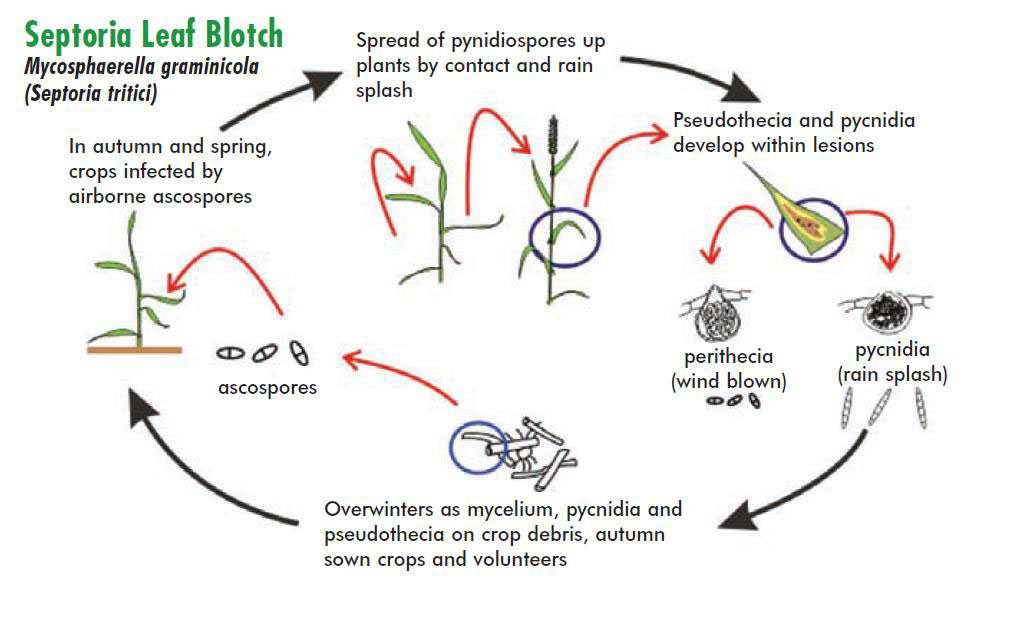
Disease-cycle of septoria leaf blotch, reproduced with permission of the AHDB, from the Encyclopaedia of Cereal Diseases.
Management Options
Fungicide choice:
See the cereals information section at The Fungicide Resistance Action Group (FRAG-UK) | AHDB or page 5 in Fungicide resistance management guide.
Generally, there has been a gradual erosion of efficacy of many azole (DMI) fungicides in the past ten years but some are still effective (e.g. propiconazole) [NB. Epoxiconazole was withdrawn in October 2021] if used at full label dose and when mixed with another mode of action. Efficacy of SDHI fungicides has declined recently but some are effective if used at full label dose alone or mixed with an azole. Resistance is widespread to QoIs (strobilurins) and MBCs (e.g. carbendazim).
Currently it is recommended to mix products with two or more modes of action or to use products with different modes of action at different spray times in order to manage fungicide resistance. Other modes of action that have reasonable efficacy when mixed with azoles or SDHIs, phthalimide (e.g. folpet), and mixtures of strobilurin and azole are also effective. [NB. Chlorothalonil is now withdrawn, mancozeb is likely to be withdrawn in 2023 or 2024].
Cultural control and climate:
Gladders et al (2001) reported that the severity of septoria leaf blotch decreased with later sowing, and with more frost days in November. In contrast, high risk “septoria” periods (rain splash events) in May and June encouraged the disease. A reduction in atmospheric SO2 concentrations in the 1970s and 1980s is thought to have caused switch from Septoria nodorum (Parastagnospora nodorum) as the main foliar pathogen of wheat in Europe to Zymoseptoria tritici in Europe (Shaw et al. 2008). Effects of climate change are difficult to predict due to contradictory effects of mild weather promoting inoculum build-up over winter but drier weather reducing infection of final leaves in late-spring (Gouache et al. 2011). The impact of the disease depends on infection of the final leaves, which is heavily affected by spring rainfall. The pathogen population is so diverse and well-dispersed that no current wheat varieties (on the AHDB recommended list) are completely resistant,
ranging from resistance ratings of 4-8.5 on a 1-9 scale where 9 is completely resistant. The most resistant varieties, Mayflower and Theodore, have resistance ratings of 8.4 and 8.5 respectively in 2022.
Gouache, D., Roche, R., Pieri P., Bancal, M.-O. (2011) section B5 in: Climate change, agriculture and forests in France: simulations of the impacts on the main species: The Green Book of the CLIMATOR project 2007-2010 part C (The Crops). Eds: Brisson, N., Levrault, F. ADEME. 336 p.
Gladders P, Paveley, Barrie, Hardwick, Hims, Langton, Taylor (2001) Annals of Applied Biology 138,301-311.
Hunter, T; Coker, RR; Royle, DJ (1999) Plant Pathology 48, 51-57.
Shaw, M. W., Bearchell, S. J., Fitt, B. D. L., Fraaije, B. A. (2008). New Phytologist 177, 229–238.